 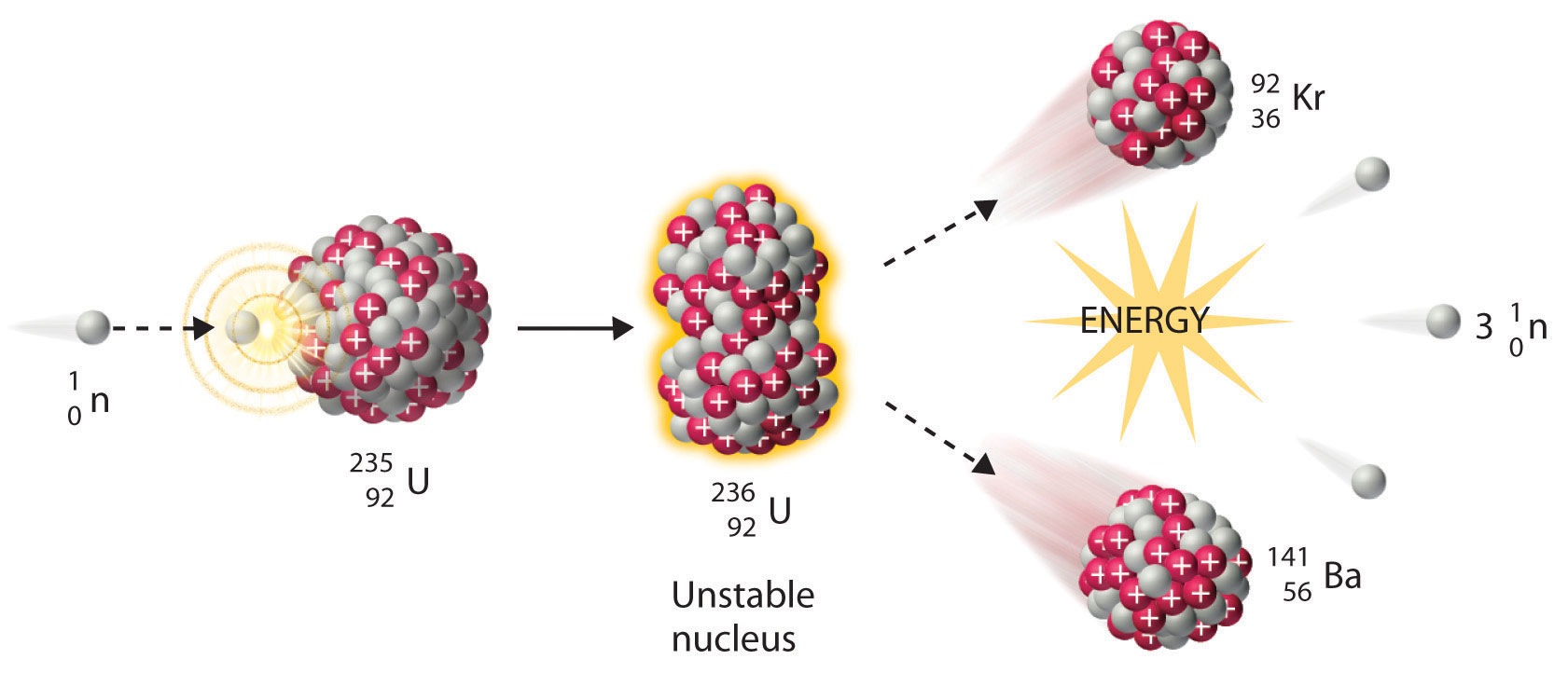
Some very heavy elements undergo fission spontaneously, while most require a nudge – something to spark the process. Nuclear fission is when a nucleus is divided in half, generating two smaller nuclei. One of the most well-known categories of nuclear reactions is the fission reaction. Types of Nuclear Reactions Fission Reactions Notice that a proton can simply be written as a hydrogen nucleus. From left: two ways of writing a beta particle or electron, two ways of writing a beta plus particle or positron, neutrino, antineutrino, neutron, and proton. Ways of representing various subatomic particles in a nuclear reaction. The total number of protons and neutrons goes just above that to produce the final written form. The number of protons is equal to the nuclear charge and goes as a subscript before the elemental symbol. The way we do this is to write each particle with a superscript and subscript to the left of its symbol.įor example, below is the symbol for lead-207, an isotope of lead with 125 neutrons and 82 protons. Sometimes we start and end with the same atoms here, so how do we show what is changing? We need to show the mass and atomic number of each nucleus or subatomic particle used in the equation. Writing nuclear reactions requires slightly different information than we are used to providing in ordinary chemical reaction equations. Most of them are only stable for fractions of a second before they decay! Writing Nuclear Reaction Equations 
Many of the synthetic elements discovered in the late twentieth and early twenty-first centuries were created by nuclear reactions. Nuclear reactions, on the other hand, modify the nuclei of atoms this can change an atom from one element into another, create different isotopes of an element, or even generate new elements that don’t exist in nature. While the chemical properties of the compounds can change drastically, the identity of the atoms remains constant in these reactions. What are nuclear reactions? Most of the chemistry we know deals with reactions between atoms and molecules. Isotope Abundance and Average Atomic Mass.How to Find Number of Protons, Neutrons, and Electrons.We also consider the different types of nuclear reactions, which include nuclear fission, nuclear fusion, and radioactive decay. This includes an introduction to nuclear reactions and a discussion about how to write their equations. In this tutorial, you will learn all about nuclear reactions. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
